Unveiling the Secrets of RNA Silencing
Prof. Tuan Anh GUYEN, Division of Life Science, and his team have published a breakthrough in molecular biology, findings that can advance gene‑regulation research into areas such as the mechanisms underlying cancer, immune disorders, and genetic diseases.
Boundless: What recent discovery has the HKUST research team made regarding the human enzyme DICER?
Prof. Nguyen: Our discovery is genuinely groundbreaking. We found that the enzyme DICER, which is crucial for gene silencing, possesses a "dual-pocket" mechanism for measuring RNA. This is significant because it changes our understanding of how DICER interacts with RNA strands.
Boundless: What exactly is “gene silencing”?
Prof. Nguyen: Good question. Gene silencing means reducing or eliminating the expression of a specific gene. This process may occur naturally in cells or be induced artificially. Gene silencing is used to prevent the production of proteins from a targeted gene. This technique helps researchers study the function of a gene, investigate disease mechanisms, and develop gene-based therapies.
Boundless: Why is DICER important in the context of gene regulation?
Prof. Nguyen: DICER plays a vital role in RNA interference (RNAi), a biological process where cells use small RNA molecules to silence specific genes. By cutting long RNA strands into smaller pieces called microRNAs, DICER helps regulate gene expression, which is essential for proper cellular function.
Boundless: What was the major innovation identified by the HKUST team?
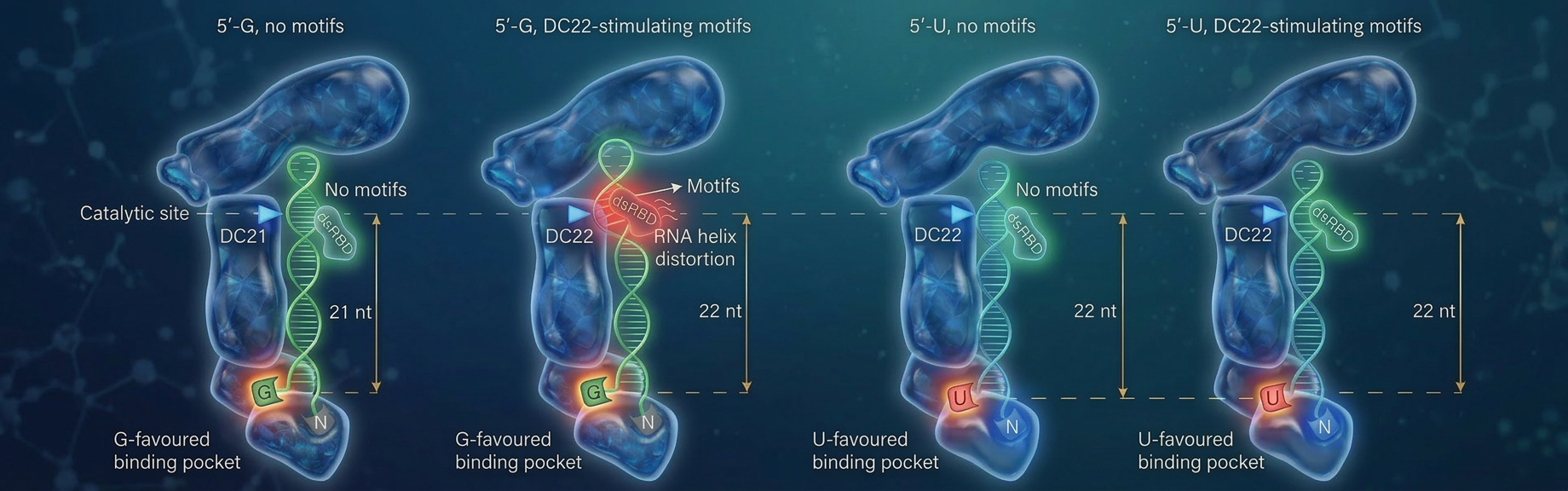
Prof. Nguyen: We have identified a previously unknown "G-favored binding pocket" in DICER that can specifically recognize RNA strands starting with the nucleotide Guanine. This finding overturns the earlier belief that DICER only had a pocket that disfavored Guanine, providing new insights into its functionality.
Boundless: How did the research team conduct their study?
Prof. Nguyen: The team employed a combination of advanced techniques, including big data analysis and high-resolution imaging. We performed parallel dicing assays that involved testing thousands of RNA variants to see how DICER interacts with them. We also used cryo-electron microscopy (Cryo-EM) to visualize the DICER enzyme holding RNA at the atomic level.
Boundless: What were some of the challenges faced during the research?
Prof. Nguyen: One of the biggest challenges was capturing the DICER enzyme in its active state while interacting with different RNA sequences. Previous studies often focused on DICER's structure when it was inactive, making it difficult to understand how it actually functions in real-time.
Boundless: What were the key findings from the study?
Prof. Nguyen: It is complex, but this is the basics. The study confirmed that the starting nucleotide of the RNA strand dictates the cut length. If the RNA starts with Guanine (G), it aligns for a 21-nucleotide cut; if it starts with Uracil, it aligns for a 22-nucleotide cut. This discovery corrected the previous theory that G-start RNAs were simply poor fits for DICER.
Boundless: What does this research mean for the future of RNA-based therapies?
Prof. Nguyen: Understanding how DICER operates is crucial for designing effective RNA-based medicines, such as short-hairpin RNAs (shRNAs) used in gene therapy. The research provides a blueprint for creating shRNAs that can target specific genes more accurately, minimizing the risk of off-target effects.
Boundless: What are the next steps for this research team?
Prof. Nguyen: The team plans to investigate how DICER processes RNA when faced with conflicting signals, such as a G-start RNA with a structural shape that demands a different cut. They also intend to apply AI and high-throughput methods to predict how DICER interacts with various RNA sequences.
Boundless: How does this discovery impact the broader field of molecular biology?
Prof. Nguyen: Another good question. Essentially, this research reshapes our fundamental understanding of microRNA biogenesis, i.e., how our bodies produce gene-regulating molecules. By providing a detailed mechanical map of DICER's functions, we can foresee future developments in RNA interference-based therapeutics, potentially leading to more effective treatments for an array of diseases.
Boundless: Thank you, Prof. Nguyen, for explaining this fascinating piece of research. The study has great promise for developing new ways of tackling health conditions. This research is led by Prof. Nguyen with his PhD students, Minh Khoa NGO and Cong Truc LE, and has been published in the prestigious journal Nature under the title “DICER cleavage fidelity is governed by 5′‑end binding pockets.”